

About Us
Manufacture
Products
Procurement
Working at Medpolymer
- Contact Us
- Ru
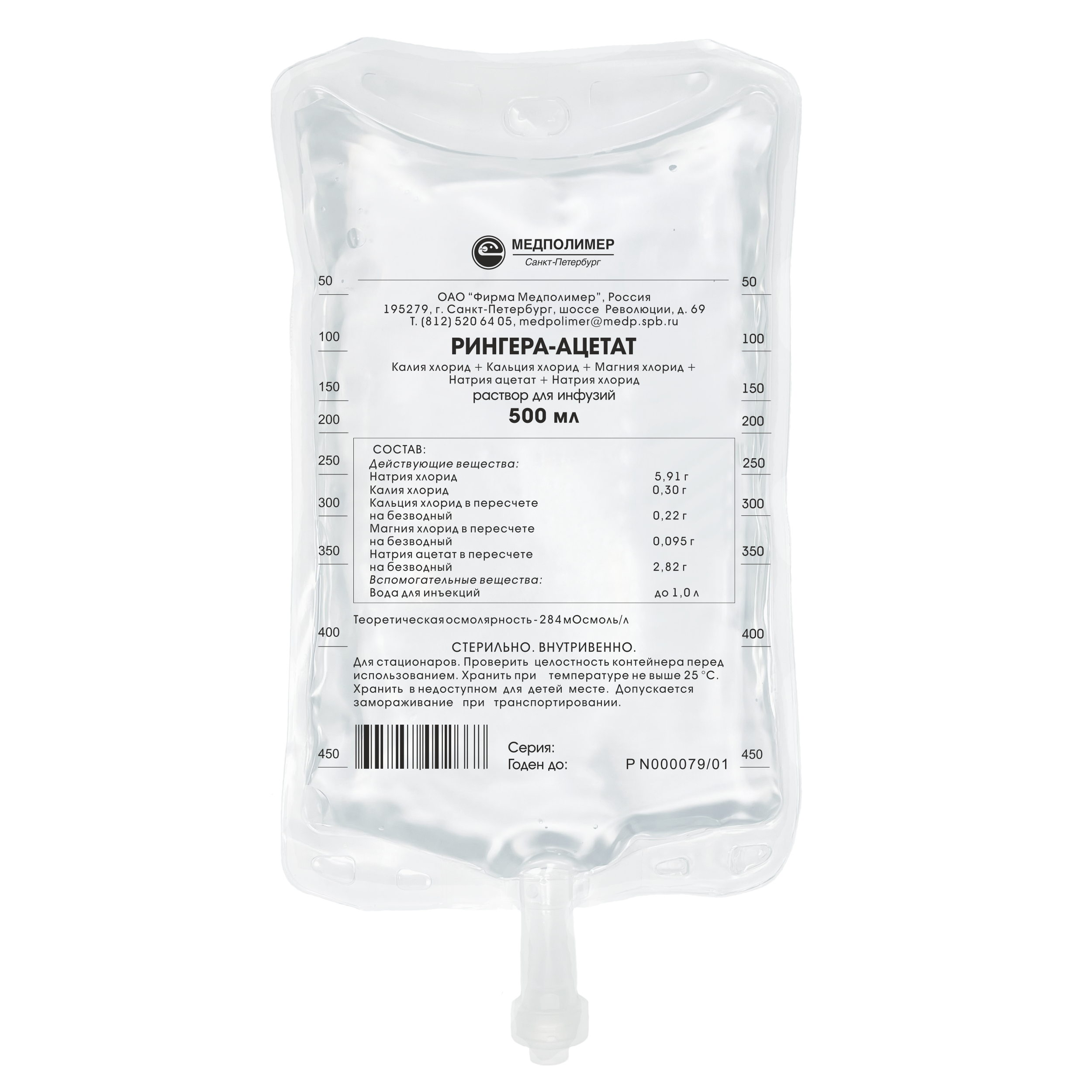
Marketing Authorisation No. P N000079/01
Composition:
Sodium chloride – 5,91 g,
Sodium acetate (on dry basis) - 2.82 g,
Potassium Chloride — 0,3 g,
Calcium chloride (on dry basis) - 0.25 g,
Magnesium chloride (on dry basis) - 0,095 g,
Water for injections – up to 1.0 L.
Theoretical osmolarity — 284 mOsm/L
Therapeutic class: electrolyte rebalancing agent
Pharmacological action: Combination drug, eliminates metabolic acidosis, regulates acid-base balance, water and electrolyte composition of blood. The therapeutic effect of the drug is associated with a temporary blood volume increase, circulating in vessels, supplementing of sodium, potassium and calcium ion deficiency, and the conversion of acetate ion into bicarbonate.
Indications: Shock, heat injury, acute blood loss; hypohydration (isotonic and hypotonic forms); metabolic acidosis; acute general peritonitis, intestinal obstruction (for correction of fluid and electrolyte balance); decompensation of electrolyte disorders in patients with intestinal fistulas; therapeutic plasma exhcange (dialysis-filter method).
Shelf life: 3 years.